Amides – Functional Group Spotlight
Amides are one of the most important functional groups in Organic Chemistry. They’re ubiquitous in biology and medicine and understanding their structure and properties is vital to understanding what gives proteins their shapes. Here with a special guest Spotlight on Amides is MedChemProf, an Associate Professor in a School of Pharmacy, with strong interest in modern technology in teaching. Read on and be sure to have a play with the cool interactive molecules scattered throughout the post! – Mark
Amides – a medal-winning functional group?
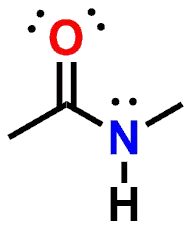
Figure 1: N-Methylacetamide
N- Methylacetamide by MedChemProf on Sketchfab
While it is difficult to argue that one functional group is more important to understand its function as compared to others, a strong case could be made for the Amide functional group to win a medal. A solid understanding of the amide functional group is critical for not only students of chemistry, but also those students planning on careers in biology, medicine, pharmacy and general science education. The reason for this is that the amide bond is a foundational building block in the overall structure of proteins, enzymes, and peptide based signaling molecules – more on peptides later.
Carboxylic acid + amine → amide + water
Let’s start with the basics of the Amide and then build our understanding from there. The Amide functional group is synthesized chemically through an overall dehydration of a Carboxylic Acid and an Amine. Once formed, the Amide is highly stable and very different in its acid / base properties as compared to its parent molecules.
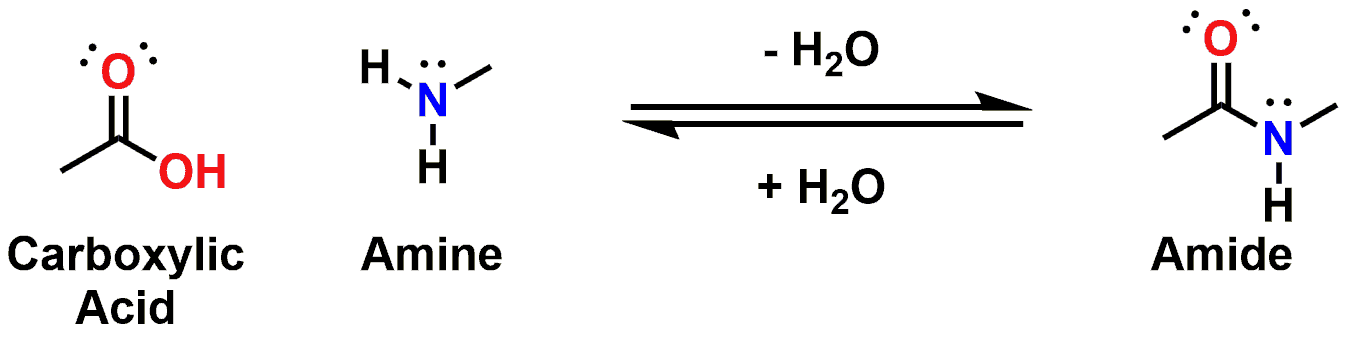
Figure 2: General process for the chemical synthesis of an Amide (Note: Biologically, peptides are predominantly synthesized in processes that involve enzymes such as Peptidyl Transferases or Peptide Synthetases. As already mentioned, we will further elaborate on peptides later in this post.)
The Carboxylic Acid precursors are acidic organic functional groups with average pKa values around 5, depending on associated substitutions. The Amine precursors, in contrast, are basic functional groups with average pKa values around 9.5, again depending on substitution adjacent to the amine. In contrast to these acidic or basic functional groups, the Amide product is considered a Neutral functionality.
Many students mistakenly assign the Amide as a Basic functional group simply because of the presence of the Nitrogen atom. However, Nitrogen atoms are only Basic when they are part of a functional group such as an Amine or Guanidine.
The nitrogen lone pair is delocalized
The reason that the Amide Nitrogen is no longer basic, as compared to an Amine, is that the lone pair of the Nitrogen is delocalized via resonance across the Nitrogen as well as the Carbon and Oxygen atoms of the carbonyl group (C=O). In order to achieve delocalization of the lone pair, the Nitrogen (as well as the Carbon and Oxygen of the carbonyl) all must adopt an sp2 hybridization state. The lone pair delocalization and sp2 hybridization also imbues the Amide functional group with a trigonal planar geometry, thereby forcing the atoms comprising the Amide and also those directly attached to it, to lie in the same plane. If you look at the 3-dimensional structure of N-Methylacetamide again, you can see that the atoms comprising the Amide, as well as those directly attached to it, lie in a single plane.
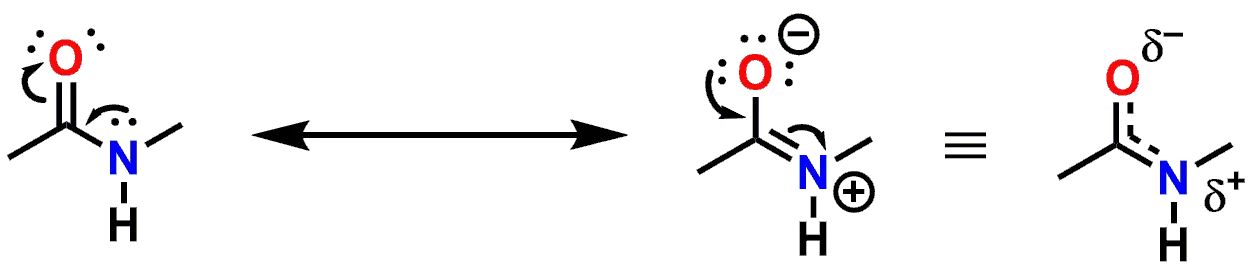
Figure 3: Resonance delocalization of the lone pair of electrons of the Nitrogen atom.
Amide-spotting
Amides can be classified as either primary (1°), secondary (2°) or tertiary (3°), based on the number of carbon atoms attached to the Nitrogen atom. If you examine the primary Amide, below, you might expect the two Hydrogen atoms to be considered equivalent if examined by NMR spectroscopy, but in fact they are non-equivalent due to hindered rotation around the bond between the Nitrogen and Carbon atoms of the carbonyl group.
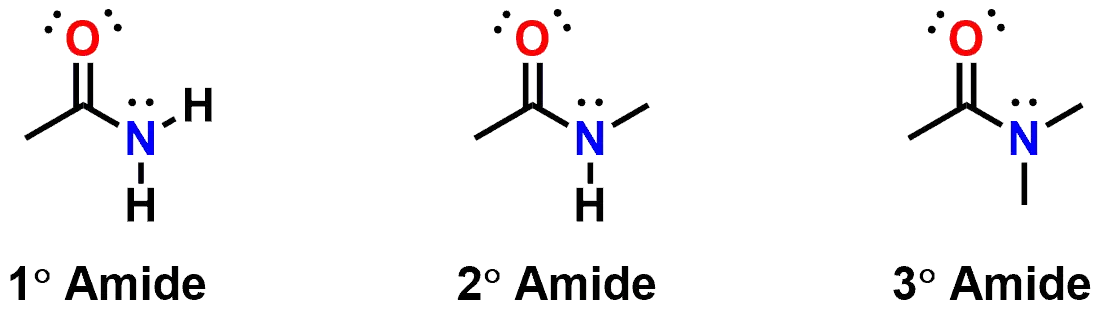
Figure 4: Classification of Amides.
While it is possible for rotation around that bond to occur, the rotation is relatively slow due to the resonance delocalization of the lone pair of electrons on the Nitrogen. The same is true of secondary and tertiary amides also. If you examine the secondary amide above, N-Methylacetamide (which is also shown in the interactive 3D representation at the beginning of this post), you will see that the molecule is drawn with the two methyl groups in a relative trans- geometry. Both the cis- and trans- conformations are possible, but energetically different. In the case of the N-Methylacetamide (secondary amide), the molecule prefers to orient the two methyl groups to be trans- to one another to reduce steric interactions.
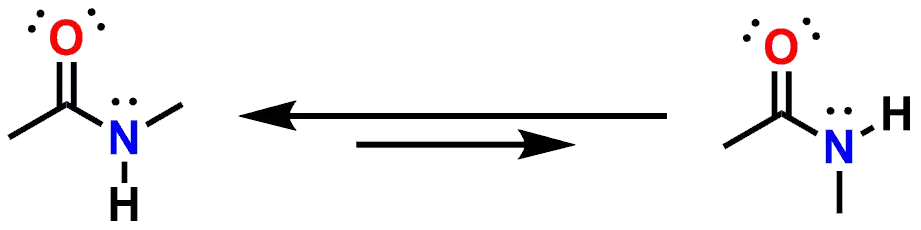
Figure 5: Equilibrium Conformations of N-Methylacetamide.
Amide functional groups play a critical role in the structure and function of proteins and enzymes. Amide linkages between the two amino acids are referred to as peptide bonds. The peptide is formed when the Amine group of one amino acid is bonded to the Carboxylic Acid group of the second amino acid.
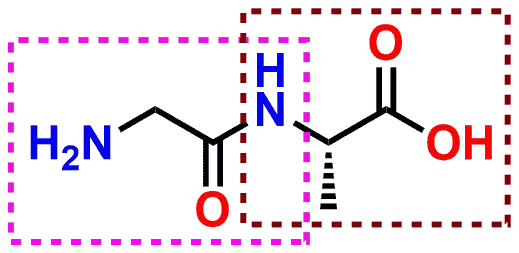
Figure 6: Example of a peptide (Amide) bond formed between the amino acids Glycine and Alanine. Note that the one Amine group and one Carboxylic Acid group remain on opposite ends of the new molecule to allow for further peptides to be added in both directions.
Peptides and polypeptides
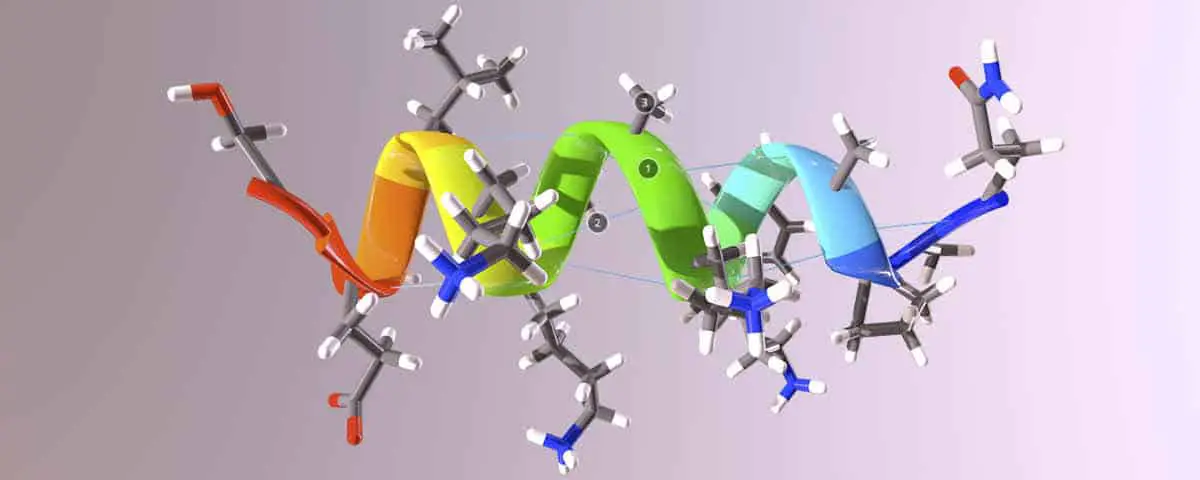
Linkages of multiple amino acids (via amide bonds) in a chain are known as polypeptides. So in reality, protein chains are actually poly-amides. The order of amino acids in the polypeptide chain is referred to as the Primary Structure (not to be confused with the term Primary Amide.) The Primary Structure of the polypeptide includes not only the linear order of amino acids, but also the conformational geometry of the individual peptide bonds (amide bonds) which is governed by the sp2 hybridization of the atoms resulting from the resonance delocalization of the lone pair of electrons on the Nitrogen. If we examine the three dimensional structure of a small protein chain shown below, you will see that each amide (peptide) adopts the trigonal planar geometry that was seen when you examined simpler amides bonds, such as with the N-Methylacetamide molecule. Furthermore, you will also see that the polypeptide also adopts an additional helical level or organization.
Secondary structure – α-helices and β-pleated sheets
Many polypeptides either adopt what is known as an α-helix or β-pleated sheet. These two levels of organization are referred to as the Secondary Structure of a protein and they result from intramolecular hydrogen bonding between the N-H of one amide and the C=O of another amide bond. These interactions are labelled on the 3D molecule shown below.
Stick Representation of a Short Polypeptide by MedChemProf on Sketchfab
Polypeptide backbones can be represented as ‘ribbons’
The helical structure of the α-helix is further highlighted using the same polypeptide where the backbone of the polypeptide (poly-amide) is now illustrated as a ribbon (below), instead of the stick representation (above). The Hydrogen Bonds are shown linking the rungs of the helical structure. You can also see the side-chains of the individual amino acids branching off from the main backbone. The ribbon representation is often used instead of the stick view in order to simplify the visualization. So now when you see a representation of a protein or enzyme, remember that the ribbon structure represents the polymeric string of amide bonds that comprise the backbone of the structure.
Ribbon Representation of a Short Polypeptide by MedChemProf on Sketchfab
Amides are also common in ‘small molecule’ drugs
While the importance of amide bonding has been made for large molecules such as enzymes and proteins, amides also comprise parts of smaller molecules. The amide functional group is relatively stable, as compared to the more easily hydrolyzed ester functional group, and is frequently present in drug molecules. If you examine the drawing of the drug Amoxicillin (below), you will notice there are two amide bonds contained in the structure. The amide labelled ‘Amide #1’ is a secondary amide. If you examine Amide #1 in the 3D Amoxicillin model, you will see that it adopts a conformation consistent with previous discussion in regards to its trigonal planar geometry.
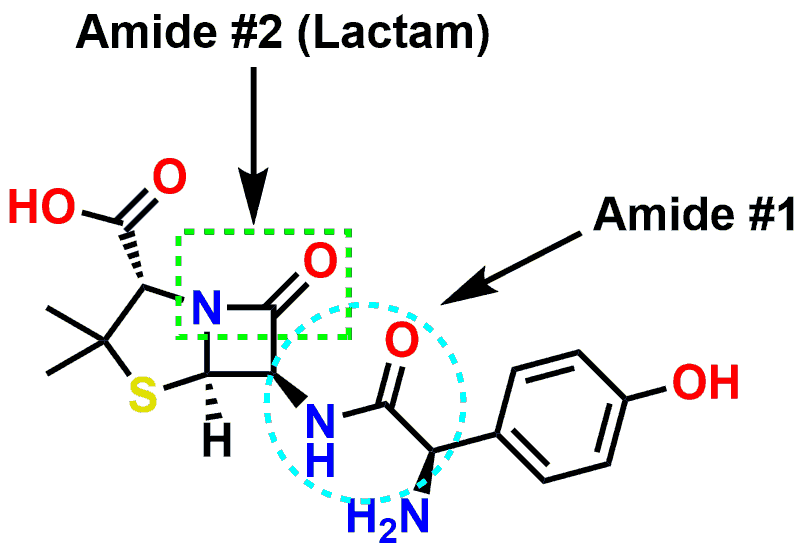
Figure 7: Amide / Lactam groups contained in the antibiotic Amoxicillin.
Amoxicillin by MedChemProf on Sketchfab
β-Lactams are 4-membered cyclic amides
There is a second amide also present in Amoxicillin which is labeled as Amide #2. This amide is somewhat special since it is part of a strained, 4-membered ring known as a β-Lactam. The term Lactam simply denotes that the amide is incorporated into part of an overall cyclic structure. The β- denotes that the ring contains 2 Carbons and cyclizes back onto itself at the second Carbon or β-Carbon (the first Carbon being the α-Carbon.) Cyclic amides (or Lactams) are usually just as stable as their acyclic counterparts. The β-Lactam of Amoxicillin is more easily hydrolyzed than a typical amide due to the increased strain from the 4-membered ring. The small ring also necessitates some deviation from the bond angles found in a normal amide (such as with Amide #1.) The β-Lactam of Amoxicillin is susceptible to hydrolysis (metabolism) from the enzyme β-Lactamase, which are commonly employed by bacteria to gain resistance to β-Lactam antibiotics.
Mark here again – I hope you found this Spotlight on Amides by MedChemProf to be interesting and useful! If you choose one functional group to really know inside and out, this would be a good choice. If you enjoyed this, be sure to also check out MedChemProf’s Spotlight on Acetals, Hemiacetals, Ketals and Hemiketals – all the ‘Al’s!
Leave a comment below if you have any questions or to suggest the next topic for a Spotlight article.





This is such a wonderful explanation of the amide functional group. Brings back happy memories of both of my graduate school days and indeed pharmacy school days too. Well done!
Thanks, I will pass your comments on to MedChemProf! It’s always great to hear “happy memories” in the context of organic chemistry!
Being a biomedical student I found this article both interesting and helpful.
Excellent! Please let me know what other topics would help you in your biomedical studies.
You guys did a great job with the 3D structures!